Along the journey of discovery, we undoubtedly carry concepts and ideas with us that we have long accepted as true. As we look closer, we may discover that some of the concepts we previously thought to be true are in reality incomplete. If this has happened to you, then welcome to the world of science and discovery. Being open to new discoveries always changes the way we perceive things. On this journey, we have seen that oxidants, traditionally thought to be bad for us, are not only good for us but they are fundamental signaling molecules in our cells and tissues. All of the evidence indicates that we need oxidants in order to live healthy lives. Let’s take a closer look at this concept and find a few examples of the things that oxidants do that make them essential to life.
According to geological records, after the formation of the earth our atmosphere did not have oxygen gas to speak of. Most of the elemental oxygen on earth at that time was tied up in water, H2O. The atmosphere was mostly composed of volcanic gasses (including carbon dioxide), water vapor and inert nitrogen. As the water vapor condensed into oceans and the earth cooled, the conditions were finally favorable for life. The earliest living organisms on earth were anaerobic (meaning that they did not require oxygen). They harnessed energy from the sun or geothermal vents to live. Some of the species of bacteria we have on earth today are still anaerobic, they do not require oxygen and also do not handle oxidants very well (oxidants are toxic to these types of bacteria).
All of the oxygen that is currently in the atmosphere has been generated by life forms on earth that break apart the water molecules and release oxygen as gas. All plant cells that perform photosynthesis utilize light energy from the sun to break apart water molecules. The sunlight captured by the plant cells provides the energy to separate two oxygen atoms from water molecules and transform them into a free radical called superoxide (O2*-), the hydrogen atoms from the water are stored for later use. In an energy-releasing cascade, the superoxide is transformed into hydrogen peroxide (H2O2) and then finally into an oxygen gas molecule (O2). The energy released from this cascade is used by the plant cells to attach the hydrogen atoms onto carbon atoms to form carbohydrates (sugars), fats and oils. These are stored and utilized as fuel. Note that superoxide, hydrogen peroxide, and oxygen produced by photosynthesis are components of the reactive oxygen species (ROS) that we call redox signaling molecules. Plants also utilize these redox signaling molecules, almost exclusively, to help detect repair and replace their own damaged cells and tissues and power their immune systems. The resulting oxygen gas produced by plant cells is then released into the atmosphere.
The redox signaling molecules produced in plant cells also help regulate the speed of production of sugars and fats inside these plant cells. Too much hydrogen peroxide, for example, is produced when the plants are in full sunlight. This excess hydrogen peroxide is designed to deactivate machinery and slow down photosynthesis, as to not overwhelm the plant. In shade, when the hydrogen peroxide level goes down, photosynthesis is sped back up. Thus redox signaling molecules are also used to regulate photosynthesis in plants.

When we eat and digest plants our body utilizes the sugars and fats we get from them as fuel. The metabolism of sugar inside our cells, combined with the oxygen we breathe, runs the same chemical process in reverse to produce energy (ATP) and also to produce redox signaling molecules (ROS) such as superoxide and hydrogen peroxide. These molecules recombine and the end products are water molecules and carbon dioxide (what the plants start with) and the grand cycle is complete. In our cells, these redox signaling molecules also help us to regulate our metabolism and help us to detect, repair, and replace our damaged cells. The advent of redox signaling molecules being produced in living cells is as old as oxygen in earth’s atmosphere. Our bodies require the oxygen, sugars and fats produced by plants.

In the field of medicine, therapies that utilize oxygen or other types of oxidants and oxidative processes are now emerging. Oxidative therapies include hyper-saturation of oxygen in the body with hyperbaric chambers, ozone therapies, hydrogen peroxide, chlorine dioxide, or therapies that stimulate oxidant (ROS) production in the cells, like pulsed electromagnetic therapies, infrared light therapies. All of these therapies have the end effect of increasing the amount of ROS produced inside living cells. Practitioners are reporting high rates of success from these therapies, especially in wound healing, tissue regeneration, and fighting infections. Exercising muscle tissue also has the effect of increasing the amount of ROS created by the muscle cells, with largely beneficial healing effects. James Watson, Ph.D. (who won a Nobel Prize for his work in the discovery of the structure of DNA) has now dedicated his life to finding ways to stimulate the production of ROS in cells, he calls ROS “the elixir of life against stressed cells” and touts that cellular ROS holds the cure for diabetes, many cancers, and dementia.
The question has been asked, is ROS an absolute requirement for the regeneration of tissue? In order to find out, researchers took a look at the regeneration of tadpole tails. Tadpoles have the ability to regenerate their tails if they happen to be cut off. Tadpoles are also partially transparent so that ROS can be seen inside the tadpole when revealed with special fluorescent dyes. It was observed that ROS is super concentrated along the growing edges of the regenerating tail. When the ROS was artificially removed by putting a strong antioxidant on the tail (vitamin C), the tail would not regrow.

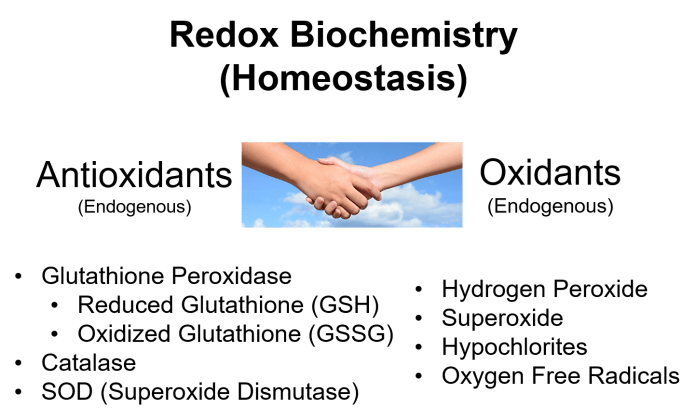
We might also ask the question, is ROS an absolute requirement inside living cells? It is now understood that in the place where proteins are manufactured inside the cell (the endoplasmic reticulum) ROS is utilized to help fold and construct the proteins correctly. Lack of ROS inside the cell has been proven to cause the proteins to be poorly constructed, ultimately resulting in disease. Proteins are the micromachines that make the cell work, they contain bonds (disulfide bonds) that can only be broken and reattached with the action of ROS. Without ROS these micromachines cannot work. The famous master antioxidant in the cell, glutathione (GSH) contains a sulfur-hydrogen thiol (SH) that is sensitive to ROS. ROS can pull off the hydrogen and causes the sticky sulfur bonds in two GSH molecules to combine to form oxidized glutathione (GSSG, the SS indicates the disulfide bond). The oxidation of glutathione when it is in its super suit (called glutathione peroxidase) requires ROS. This is true for other major actors in the cell, like thioredoxin. Without ROS, these super machines cannot operate. So it turns out that ROS, when in homeostatic balance, is absolutely essential to the correct operation of the cell in many areas and in many occasions, especially in the machinery that repair and regenerate cells and tissues. It was surprising to discover how important ROS is important in all aspects of cell life, from photosynthesis to metabolism of sugars, detection, repair and replacement of damaged cells, regeneration of tissue, immune activation, and the production of proteins.
My own work in the last 10 years has been to find a way to put a safe form of balanced, stabilized ROS (containing many of these redox signaling molecules) into a bottle, observe the results in studies and to educate the world on the science. This has been to date the most satisfying work of my career. The possibilities for these redox signaling molecules seem to be endless.

Magific Learning Lesson!..Thank yoou Dr.!
Dr. Samuelson This gives the answer to why athletes and people who exercise or seeking extreme benefits with ASEA, I LOVE THIS
Shawn Burke Founder and CEO 1950 Bellerive Lane Suite 106-A Coeur d’Alene, Idaho 83814 (208) 660-9378 http://www.withperformance.com
>
Thank you. I always look forward to your writings. Thank you for sharing your knowledge.
Once again Dr Gary you are more than helpful with your information and for this I thank you.
Thank you Dr Samuelson for a great explanation. Your work is definitely advancing life for humanity, so grateful for ASEA and your brilliant discovery…GOD BLESS.
Dios lo bendiga por el maravilloso desarrollo científico al desarrollar el proceso de estabilizar las moléculas Redox . Maravillosa su explicación. Cada día me enamoro más de ASEA , tsetvirba la humanidad con esta ciencia es una obligación que tengo como misión. Sus enseñanzas las subo en mi canal de YouTube . Conny Luque . Vamos por Sur America desde Colombia .
Peoples over use of antioxidants can cause them many health problems. Reducing H2O2 levels too low can inhibit the bodies ability to produce energy. The thyroid needs H2O2 (hydrogen peroxide) to produce thyroid hormones. Low H2O2 levels will cause low thyroid hormone levels. Balance is key to healing but many keep taking supplements after they heal instead of getting their nutrients from food causing new health problems. Also the body needs a certain level of oxidative stress to break down proteins in the body and to fight infection. By over use of antioxidants they can start to inhibit their immune system and the bodies ability to heal. Balance is key. Supplements are good when someone is chronically ill to help the body get the upper hand but once a person has healed they should try and get as many of their nutrients from food as they can.